.
How Does Cannabis Affect the Immune System?
Early research shows that cannabis could suppress the immune system. While this seems promising for diseases caused by an overactive immune system, it could result in an increased risk of infection in healthy people. However, the understanding of how cannabis influences immune function remains murky. Find out what the studies say below.
Contents:
We wouldn’t last very long without our immune system. We’re constantly surrounded by bacteria, fungi, and viruses that would jump at the chance to access the resources within our cells. Our immune system comprises numerous organs, cell types, and proteins that provide several lines of defence against these external threats. However, they aren’t always enough to stamp out an infection before it takes hold. We all encounter colds, flu, and other infectious illnesses every now and then, but the genius of our immune system ensures that we’re better equipped to deal with them next time.
In order to reduce the likelihood of getting sick, many people look for ways to boost or modify their immune system through nutrition, exercise, lifestyle adjustments, and supplements. While science supports some of these strategies, others are treated with more scepticism. But where does cannabis autumn on this spectrum? Can the herb help to bolster our cellular defences and prevent or minimise infections? Or does it make matters worse? Below, we explore these questions and more.
How the Immune System Works
Before we get into how cannabis could impact immunity, let’s quickly cover how the immune system works. Our physiological defences feature two primary categories: innate immunity and adaptive immunity.
We are all born with an innate (or general) immune system that serves as the first line of defence against pathogens entering the body. It comprises barriers such as the skin and the mucous membranes (the inner lining of the nose, mouth, lungs, and other organs and cavities) that physically prevent the movement of pesky germs.
These biological walls also harness enzymes, acids, and mucus to discourage the formation of bacteria and viruses. Scavenger cells known as phagocytes also make up the innate immune system. “Phago” stems from the Greek “phagein”, which means “to consume”. Phagocytes live up to their name by enveloping and “eating” intruding pathogens.
Whereas our innate immunity uses a scattershot and unselective strategy to destroy invaders, our adaptive (or acquired) immunity works much more specifically to tackle intruders. If our innate defences fail, the adaptive immune system arrives as backup and begins to identify the pathogen and create specific antibodies designed to kill it.
Below are the two main players involved in this process:
| T lymphocytes | These cells activate other immune cells, detect and destroy cells affected by viruses, and form “memories” of pathogens to ensure future immunity. |
| B lymphocytes | Forged in the bone marrow, these cells transform into plasma cells and churn out large numbers of antibodies—compounds made from sugars and proteins that are specifically designed to attach to and destroy an antigen. |
| T lymphocytes |
| These cells activate other immune cells, detect and destroy cells affected by viruses, and form “memories” of pathogens to ensure future immunity. |
| B lymphocytes |
| Forged in the bone marrow, these cells transform into plasma cells and churn out large numbers of antibodies—compounds made from sugars and proteins that are specifically designed to attach to and destroy an antigen. |
Cannabis, the Endocannabinoid System, and Immunity
The immune system doesn't work in isolation—nothing in the body does. If you know a thing or two about cannabis, you’ve probably heard of the endocannabinoid system (ECS). Researchers came across the components of this system while studying the effects of cannabis on the body. Eventually, they figured out that its components show up all over the body, from the brain and bones to the skin, digestive system, and immune system. They deemed the ECS the “universal regulator” of the human body, as it helps to keep everything in a state of balance, otherwise known as homeostasis.
The classical ECS features two receptors (CB1 and CB2), endocannabinoids that act as signalling molecules (anandamide and 2-AG), and enzymes that build and break down endocannabinoids. These components are also found throughout the immune system, where they help to control immune function, drive homeostasis, and modulate the immune system. A wide array of immune cells feature CB1 and CB2 receptors, including B cells, natural killer cells, monocytes, and CD8 and CD4 lymphocytes. Endocannabinoids bind to these sites and help to regulate processes such as the inflammatory response[1].
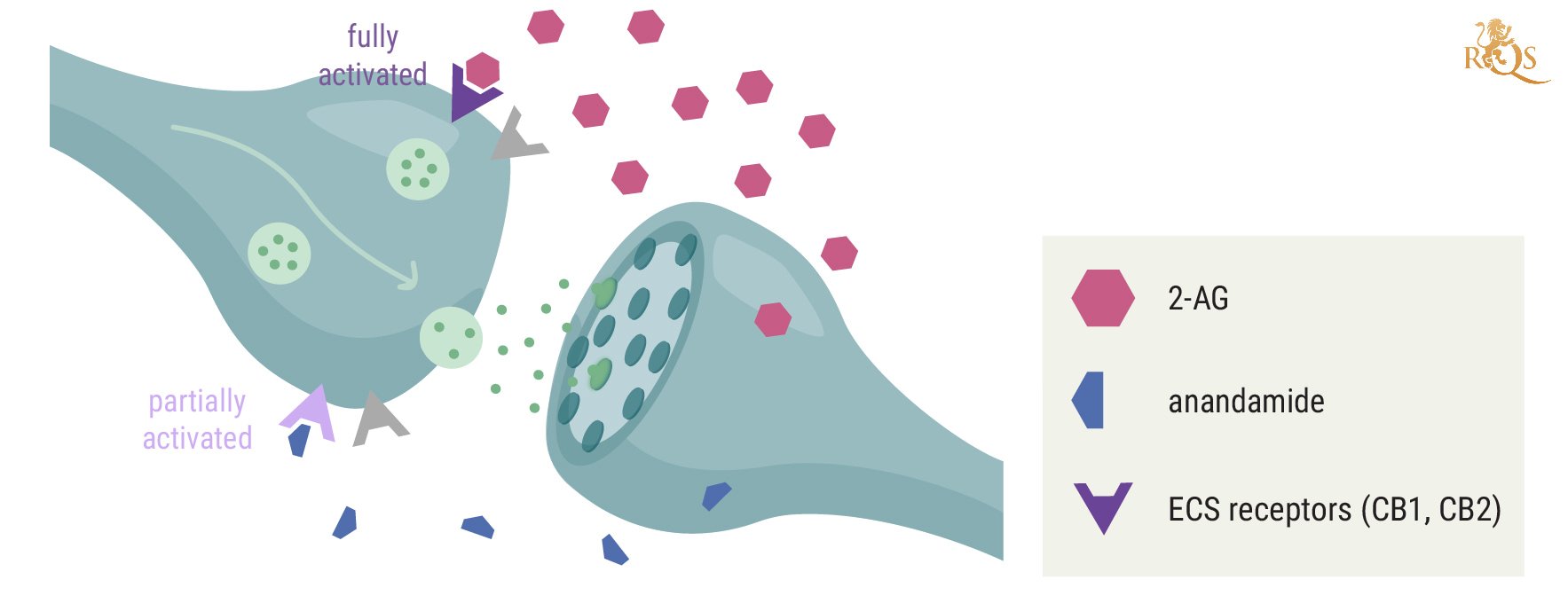
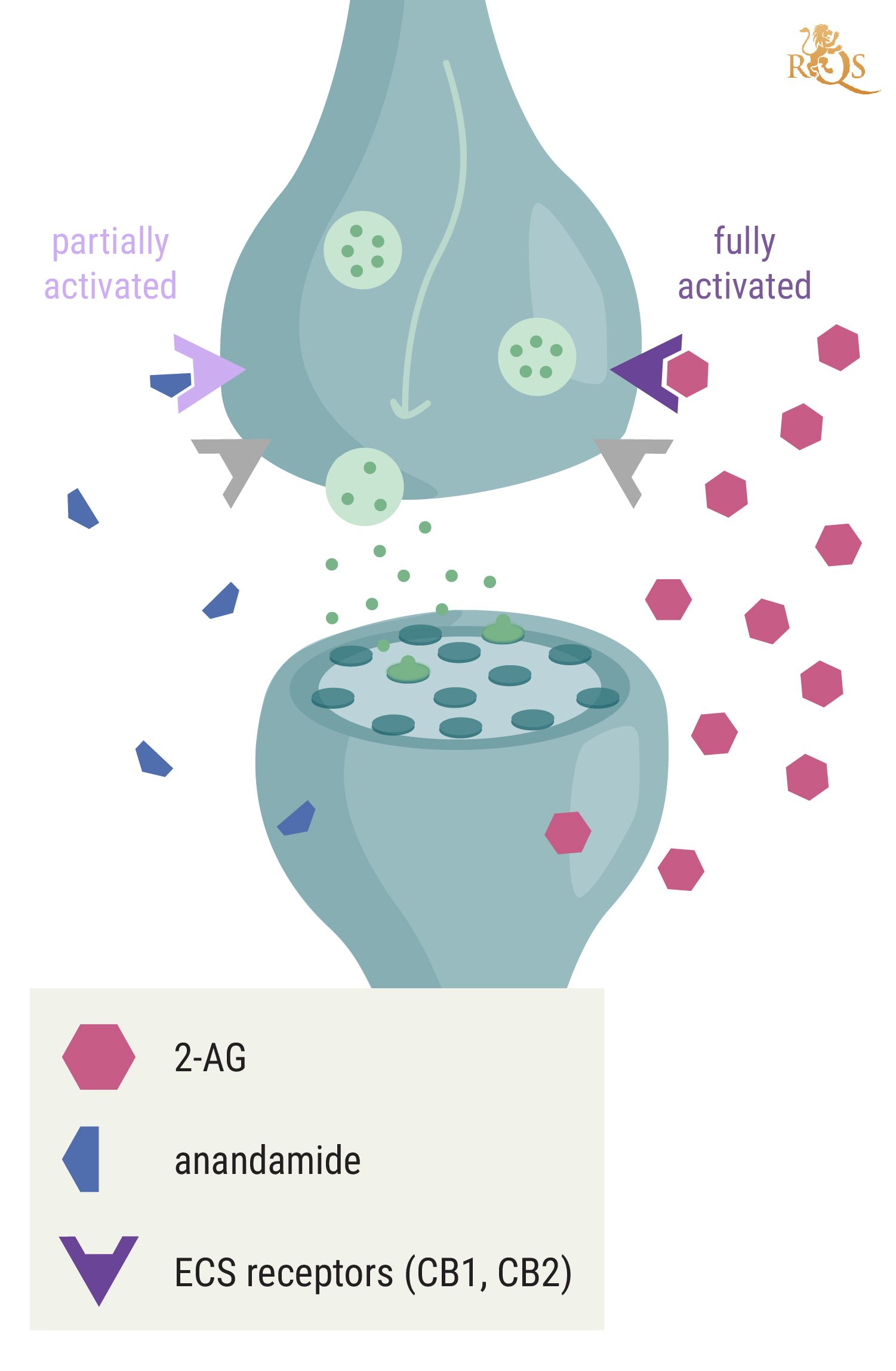
Can Smoking Weed Weaken the Immune System?
Some cannabis users, especially those with overactive immune systems, consume the herb in an attempt to tame their symptoms. However, emerging research suggests that cannabinoids—a large group of constituents found in the plant—could also dial down immune function in healthy populations. In theory, a dampening of the immune system following cannabis use could predispose users to respiratory infections.
Researchers have only scratched the surface regarding the interactions that take place between cells and receptors of these systems and the plethora of compounds found within cannabis flowers. Cannabinoids such as THC and CBD certainly influence the immune system, but exactly how they do so remains unsolved.
Preliminary investigations[2]suggest that cannabinoids modulate immune function throughout the body, from the periphery to the brain. Researchers are looking to see if these compounds can influence T cells (a type of white blood cell that fights infections) and cytokines (communication molecules secreted by immune cells).
Ongoing studies are assessing the use of cannabis for autoimmune conditions. This group of diseases, including rheumatoid arthritis and lupus, are caused by a malfunction within the immune system that directs cells to attack tissues within the body. Researchers are exploring the potential mechanism of action[3] of cannabinoids to reduce inflammation and immune cell activation to protect against this self-inflicted damage.
Although helpful in certain clinical scenarios, if cannabinoids do prove to reduce immune cell activation, this opens up the possibility that these compounds could compromise our bodily defences under normal circumstances, increasing the risk of impaired immune function[4] and respiratory diseases.
CBD and the Immune System
So, where does cannabidiol stand when it comes to weed and the immune system? Does CBD help the immune response? Or does it play a role in lowering its activity? Unlike THC, CBD doesn’t have much affinity for the primary receptors of the endocannabinoid system. However, preliminary research shows that the molecule might elevate endocannabinoid levels.Little evidence claims that CBD works to boost the immune system, but ongoing studies are looking to determine the immunosuppressive propertiesof the cannabinoid.Thorough human trials are needed to determine if CBD can produce notable effects in patients with autoimmune conditions.
How Cannabis Affects the Immune System
The intimate link between the ECS and the immune system opens up the possibility of cannabis working as a modulating agent of our physiological defences. You see, endocannabinoids (those found in the body) and phytocannabinoids (those found in plants) share a similar structure. This means that external cannabinoids, including THC and CBD, are potentially able to bind to ECS receptors, influence enzyme activity, and generally mimic endocannabinoids. As our endocannabinoids hold significant influence over our immune system, plant cannabinoids might serve as a way of “hacking” the ECS when it comes to immunity.
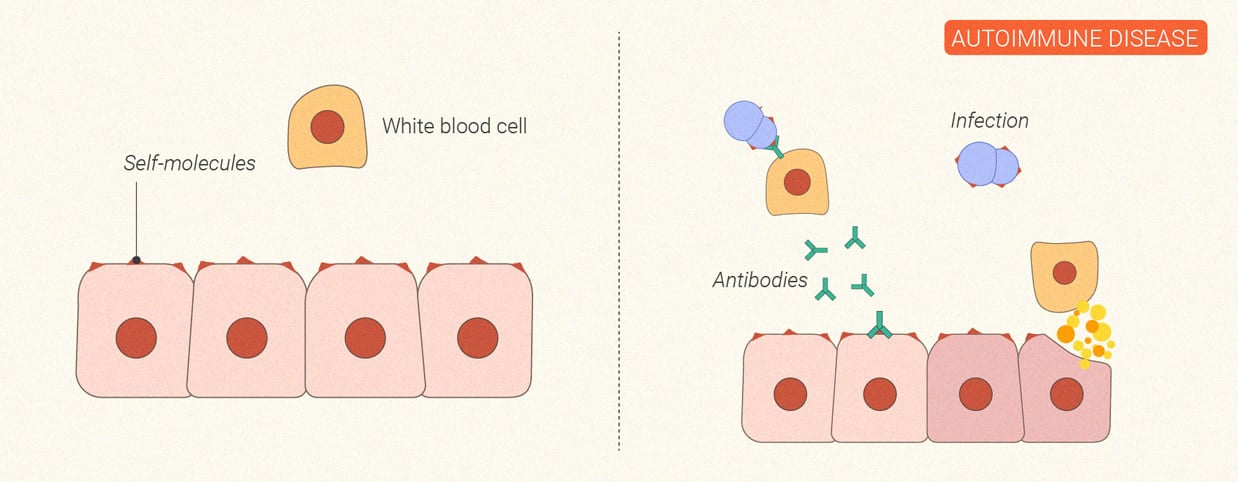
Autoimmune Disorders
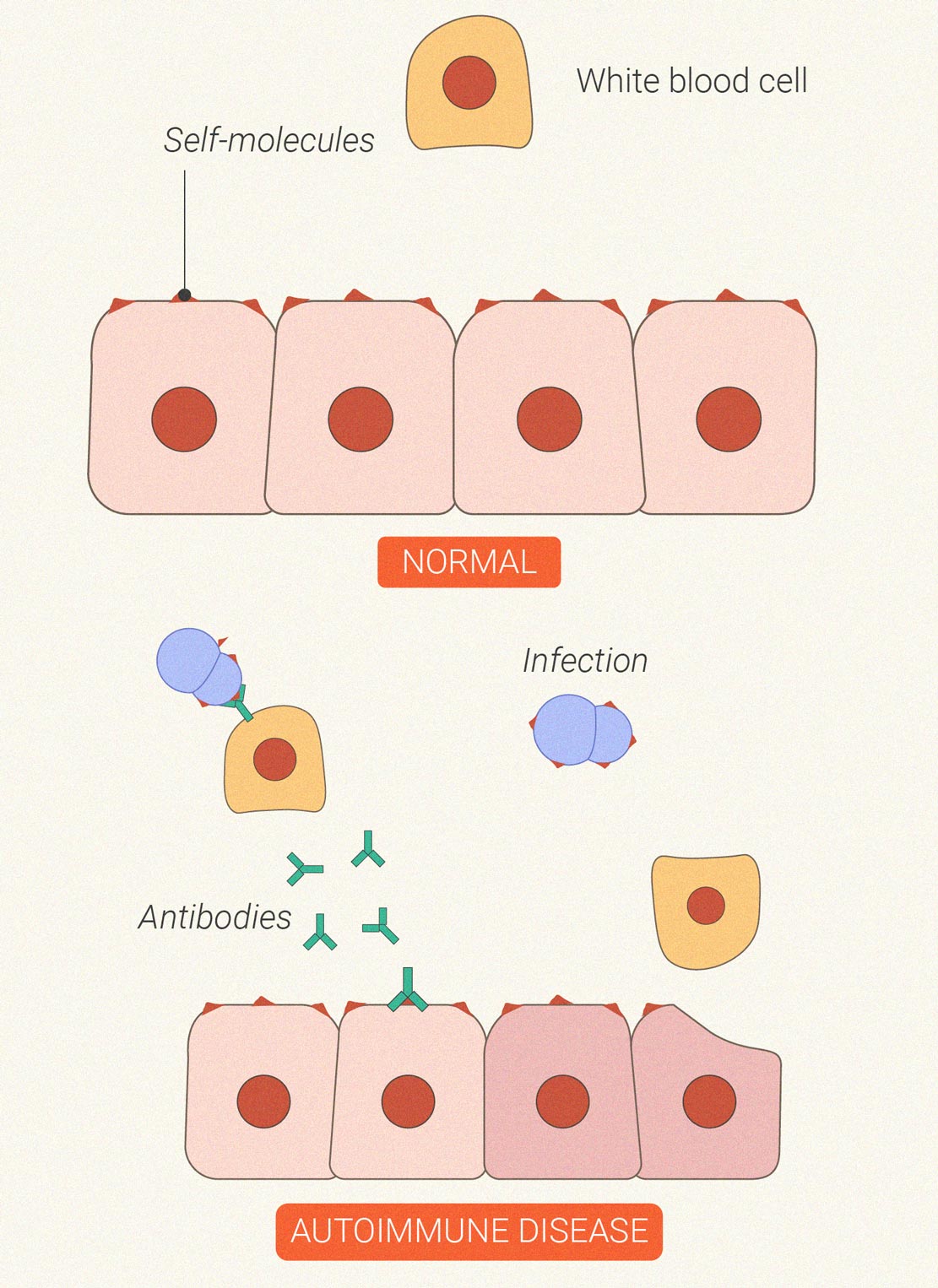
Sometimes the immune system goes haywire. In the case of autoimmune diseases, the cells that are supposed to protect us from outside invaders start to turn on our own bodily tissues; they mistake joints, skin, and nerve cells for aggressive bacteria and viruses. This self-inflicted damage leads to inflammatory cascades that result in symptoms such as fatigue, achy muscles, fever, hair loss, and rashes. Common autoimmune conditions include rheumatoid arthritis, psoriasis, and multiple sclerosis.
Early research has pitched cannabinoids against inflammation involved in autoimmune disease progression. Animal and cell studies also suggest that cannabis could exert an immunosuppressive effect.
Cannabis and Multiple Sclerosis
Multiple sclerosis (MS) also autumns into the autoimmune category of diseases. The condition gives rise to symptoms of fatigue, pain, and muscle spasms as immune cells begin to attack the brain and nerves. The exact cause of MS remains unknown, but a combination of genetic and environmental factors is likely to blame. Researchers are eager to find out if cannabinoids can benefit the condition in any way, from neuroprotection to reducing immune activation. The medical advisors of the MS Society also believe that medical cannabis could help around 1 in 10 patients when it comes to muscle spasticity.
The spasticity associated with MS can also result in an overactive bladder, causing increased frequency and urgency of urination. Ongoing studies are looking to see if cannabinoids could help patients in this area. However, the results are mixed so far. Cannabinoid receptors certainly play an important role[5] in the urinary bladder, but research published in The American Journal of Medicine suggests that cannabis use may increase the risk[6] of an overactive bladder. Despite this data, other studies continue to assess the effects of cannabis extracts[7] on incontinence in patients with MS.
Risk of Viral Infection and Immunosuppression
If cannabis interacts with the immune system in a way that suppresses it, could it give viruses and other infectious pathogens an upper hand? It’s a possibility. Because of this effect, cannabis could predispose long-term and heavy users to an increased risk of acquiring and transmitting infections, and impaired immune function in general.
Even more worrisome, limited research also suggests that long-term cannabis use could trigger myeloid-derived suppressor cells (MDSCs), which suppress the immune system and may increase the odds of cancer. However, despite their effects on the immune system, ongoing studies are exploring some cannabis compounds for their impact on pathogenic viruses and bacteria.
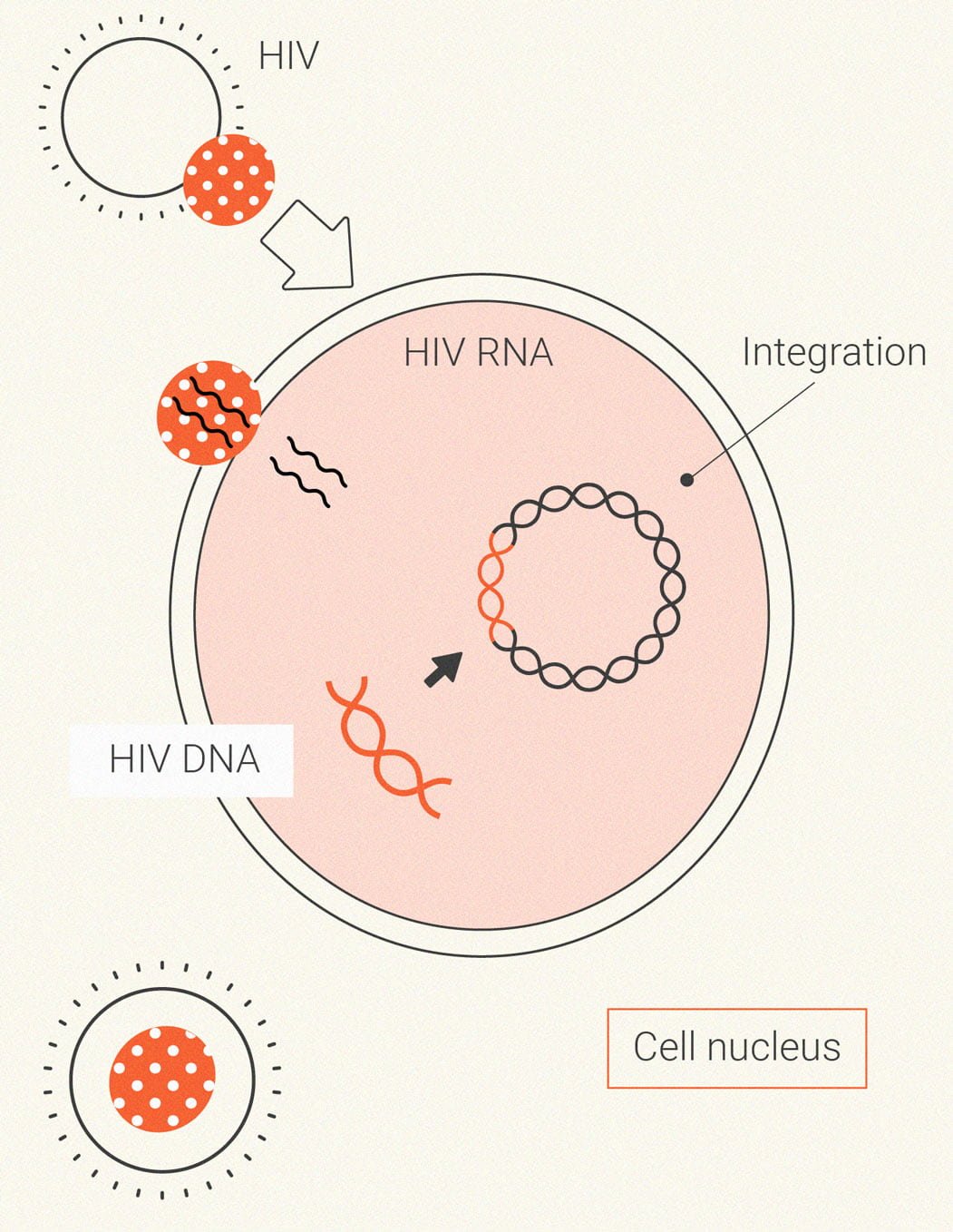
Cannabis and HIV
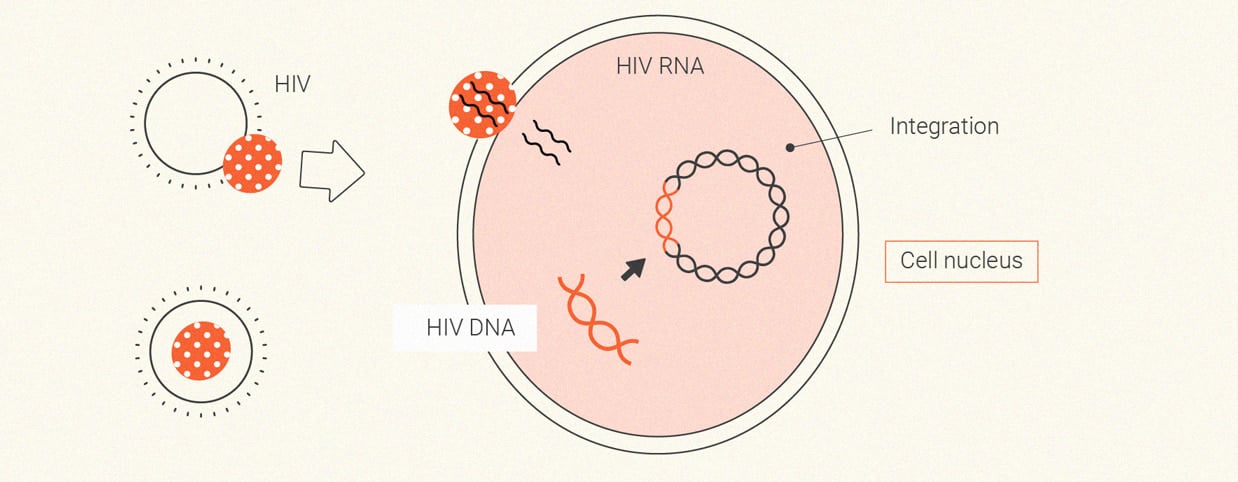
Human immunodeficiency virus (HIV) primarily spreads through unprotected sex. Once it enters the body, the pathogen acts as a parasite that lays waste to the immune system. In the early stages of infection, HIV causes flu-like symptoms and a significant reduction in CD4+ T cells that help to recruit other immune cells when faced with an infection. Following these initial symptoms, the virus continues to replicate but may not cause further symptoms for several years. Under the surface, however, it continues to impair and weaken the immune system, eventually causing a person to become immunocompromised.
Because researchers are exploring the immunosuppressive effects of cannabis, it seems logical that people living with HIV should avoid the plant. However, surveys show that many of them use the herb[8]. As well as trying to figure out the dangers of cannabis in HIV patients, scientists are also seeing if cannabinoids can help to reduce viral replication and improve T cell counts[9].
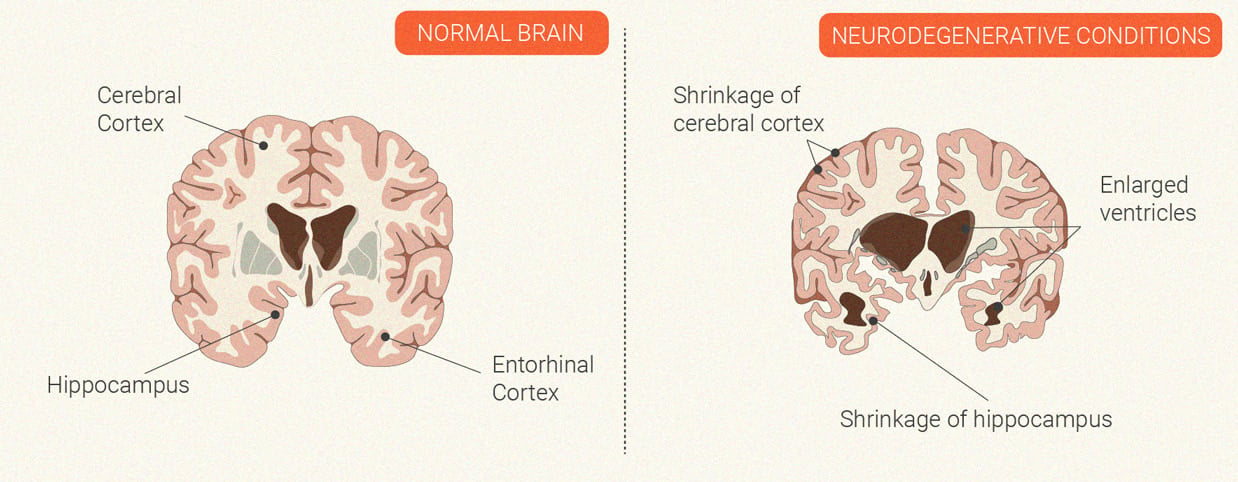
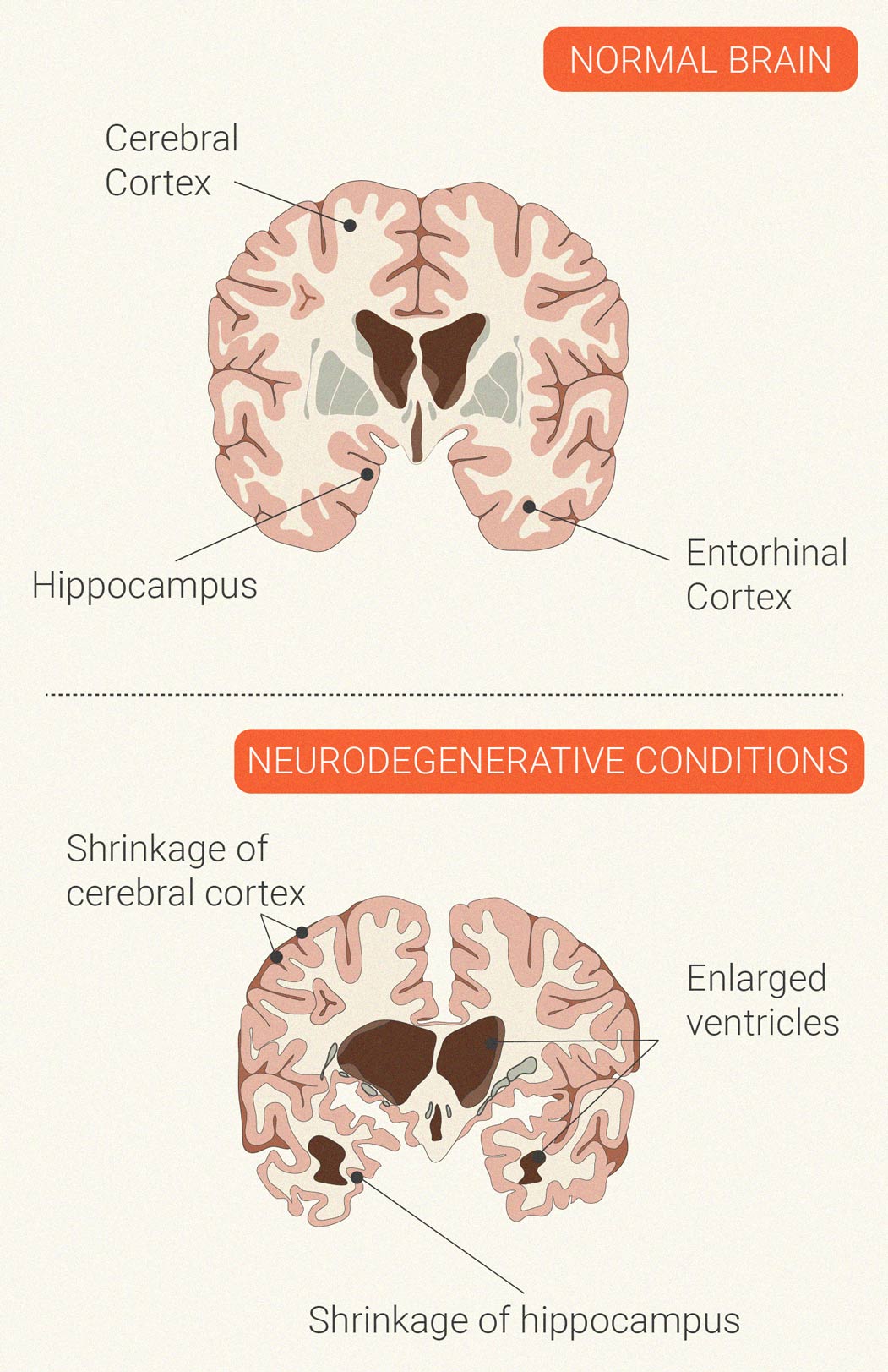
Neurodegenerative Conditions
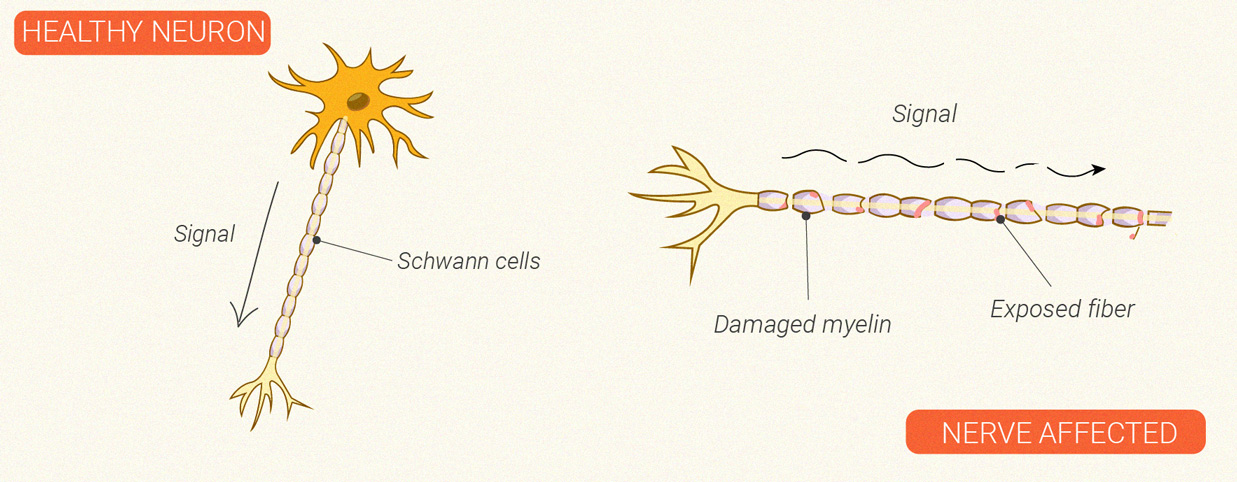
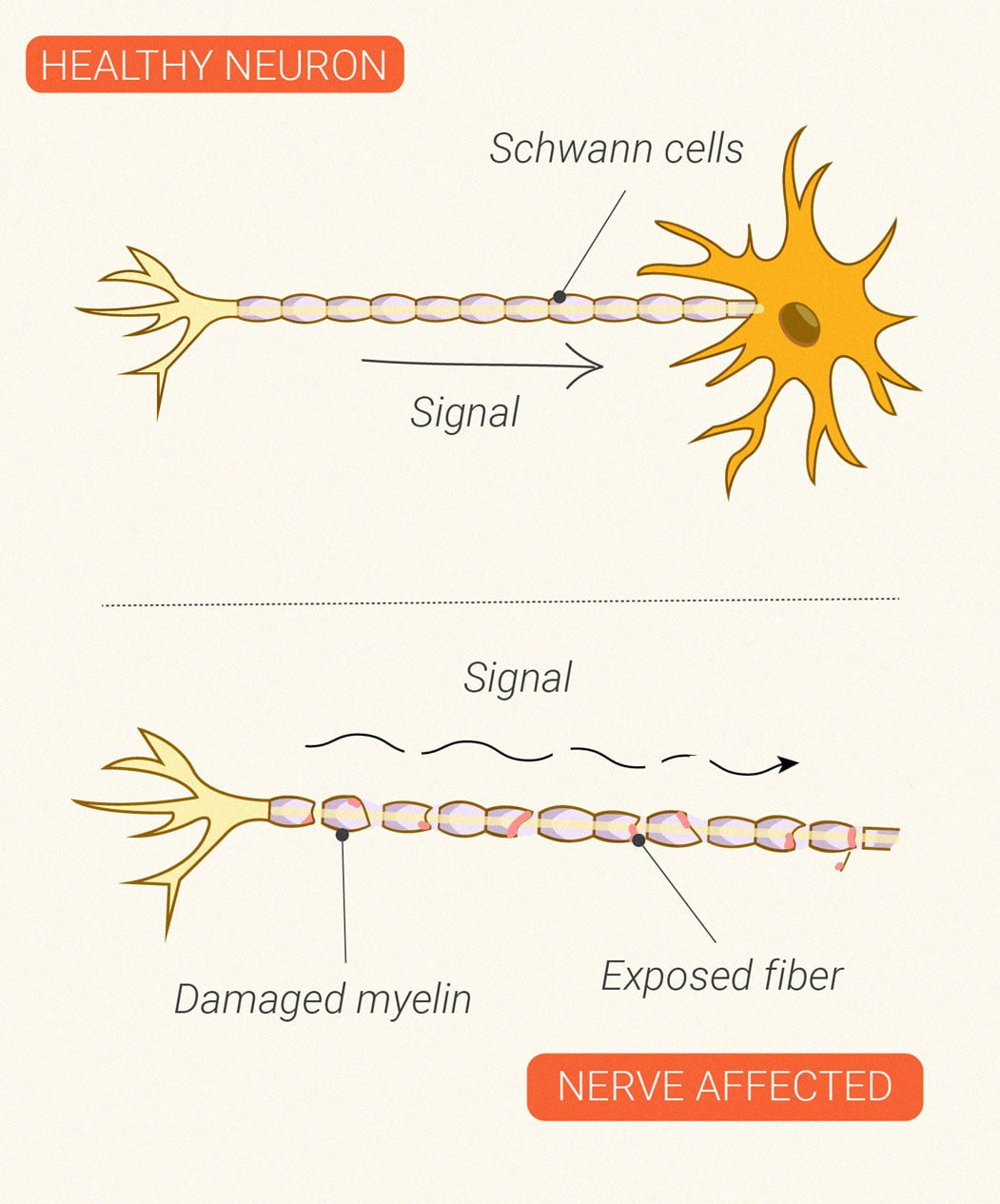
The autoimmune disease multiple sclerosis is characterised by neurodegeneration. Immune cells launch an inflammatory attack against the central nervous system. During this process, the cells develop a particular affinity for myelin—the protective and insulating coating that encases nerve cells. Over time, this assault damages the myelin and the nerve itself, which can cause problems with nervous system firing. Ongoing studies are looking at a potential neuroprotective effect[10] of the CB1 receptor and compounds that bind to this site.
Cannabis and ALS
ALS, short for amyotrophic lateral sclerosis, is a progressive neurological disease that targets the brain, nerves, and spinal cord. The early stages of the disease cause weak, tight, and spastic muscles. Sadly, the condition eventually progresses toward fatal paralysis. The exact cause of ALS remains unknown; some researchers believe it has an autoimmune aspect, whereas others reject this theory. However, research published in the journal Frontiers of Neurology found alterations in immune cell counts and cytokine levels in patients with the disease. This suggests that immune activation plays a role in a subgroup of ALS patients.
Currently, researchers are examining cannabinoids as possible therapeutics for ALS. In particular, they’re testing the compounds for their potential to reduce neuroinflammation, excitotoxicity (toxicity caused by the excessive firing of neurons), and oxidative damage. The ability of cannabis to suppress elements of the immune system could also prove helpful if future studies determine a solid correlation between autoimmunity and ALS.
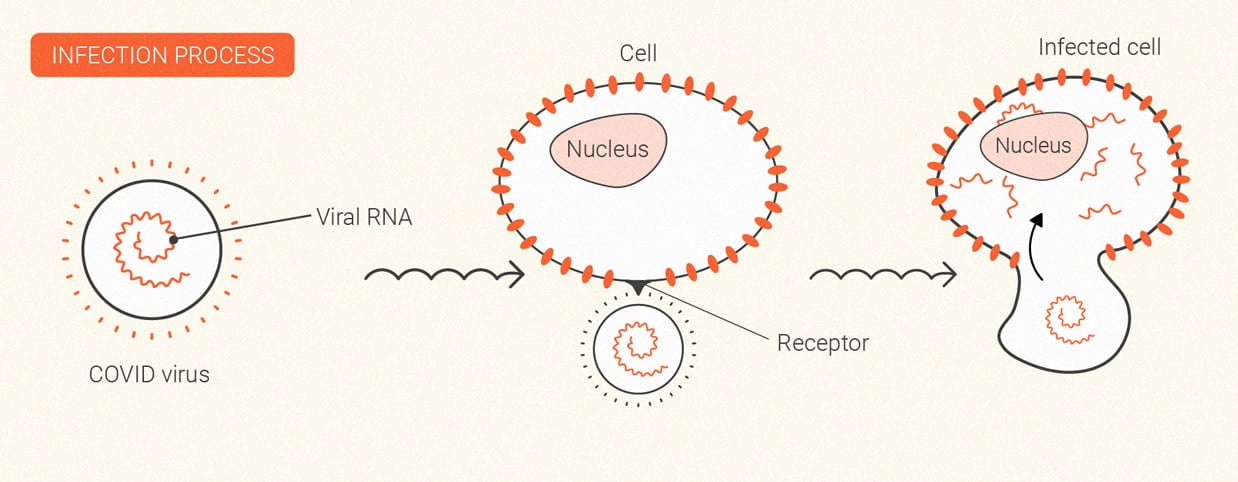
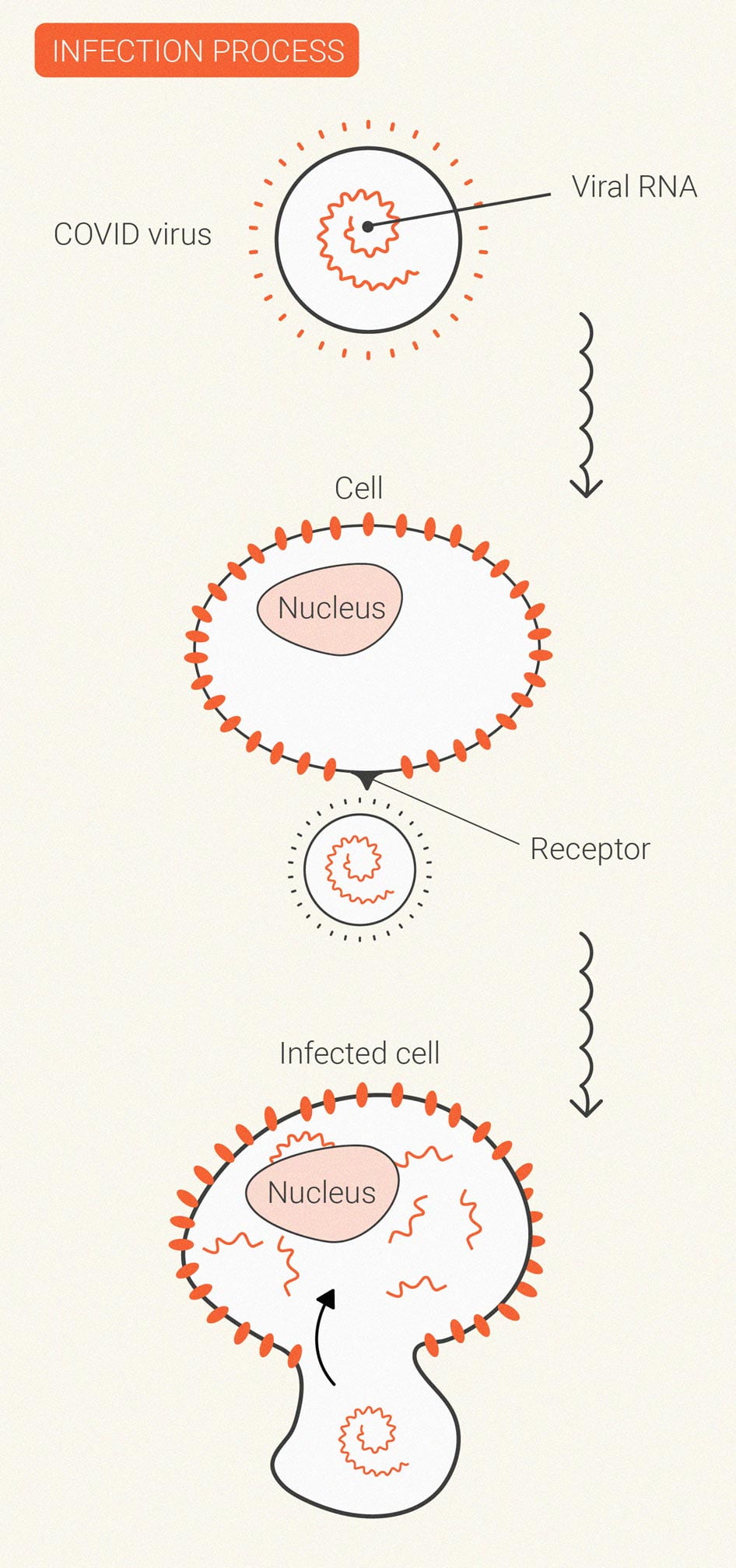
Coronavirus
SARS-CoV-2, which causes the disease COVID-19, changed the world. Despite the worldwide vaccine rollout, cases continue to rage in many regions, and researchers are still on the hunt for therapeutics.
Some research teams have decided to probe cannabis for potentially useful molecules. Although certain cannabinoids may dial down the immune response, others are being explored for their direct effect on virus particles. Additionally, the deadlier end stages of COVID-19 are driven by inflammatory storms.
Currently, researchers from Portugal are testing blends of CBD and terpenes[11] against SARS-CoV-2 infectivity. Other researchers are testing[12] the cannabinoid acids cannabigerolic acid (CBGA) and cannabidiolic acid (CBDA) against SARS-CoV-2 entry into host cells.
Is Cannabis Good or Bad for the Immune System?
We don’t have enough data to provide a confident answer to that question. Some evidence suggests that the herb has immunosuppressive properties that could be beneficial; however, if this is true, it could also potentially cause problems in those with compromised immune systems, as well as healthy individuals that use the herb often. The bottom line: we need more human trials to ascertain a clear-cut answer.
- Endocannabinoids and immune regulation - PMC https://www.ncbi.nlm.nih.gov
- Cannabinoids and the immune system: an overview - PubMed https://pubmed.ncbi.nlm.nih.gov
- Cannabis and Autoimmunity: Possible Mechanisms of Action - PMC https://www.ncbi.nlm.nih.gov
- The Link between Cannabis Use, Immune System, and Viral Infections - PMC https://www.ncbi.nlm.nih.gov
- Cannabidiol for neurodegenerative disorders: important new clinical applications for this phytocannabinoid? - PMC https://www.ncbi.nlm.nih.gov
- Cannabinoids and autoimmune diseases: A systematic review - PubMed https://pubmed.ncbi.nlm.nih.gov
- About cannabis and MS | MS Society https://www.mssociety.org.uk
- Functional role of cannabinoid receptors in urinary bladder - PMC https://www.ncbi.nlm.nih.gov
- Relationship Between Marijuana Use and Overactive Bladder (OAB) https://www.sciencedirect.com
- The effect of cannabis on urge incontinence in patients with multiple sclerosis: a multicentre, randomised placebo-controlled trial (CAMS-LUTS) - PubMed https://pubmed.ncbi.nlm.nih.gov
- The Safety and Efficacy of Marijuana in Persons Living with HIV - PubMed https://pubmed.ncbi.nlm.nih.gov
- Cells | Free Full-Text | Cannabinoids Reduce Extracellular Vesicle Release from HIV-1 Infected Myeloid Cells and Inhibit Viral Transcription https://www.mdpi.com






































