.

Cannabis terpenes: Nerolidol—A Versatile Sesquiterpene
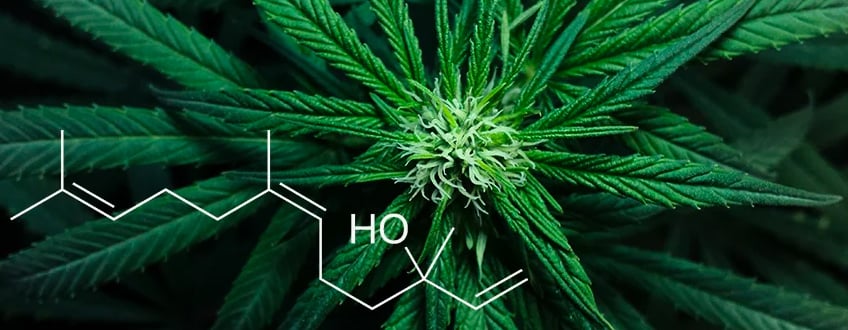
Nerolidol is found in ginger, jasmine, tea tree, and other plants, cannabis included. With its woody and fruity scent, it's used in food and essential oils, but it has medicinal properties too! Discover what research has to say about the properties of this sesquiterpene.
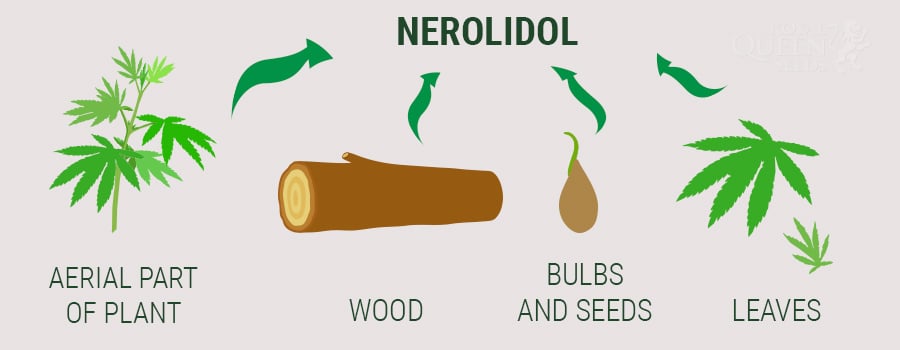
Nerolidol is a sesquiterpene, which means it is less volatile and more aromatic than monoterpenes and other evaporative substances. Several plants contain nerolidol, often found in their essential oils. These plants include ginger, jasmine, tea tree, lavender, and lemongrass, all of which are frequently used in aromatherapy.
Nerolidol is also found in large quantities in Aframomum pruinosum, Myrceugenia cucullata, Siparuna guianensis, Melaleuca quinquenervia, Piper claussenianum, Eucalyptus nova-anglica, Brassavola nodosa, and Salvia runcinata. Like other terpenes, nerolidol is one of the substances produced by plants in response to insect attacks. This terpene smells earthy and woody, yet with fruity, floral notes of citrus, apples, and rose. It’s a minor component in the terpene profiles of many cannabis strains.
Nerolidol is a big enemy of many bacteria, fungi, parasites, spider mites, head lice, etc. However, humans can safely ingest and inhale it. It has been traditionally used for its relaxing effects, and it’s commonly used as a food-flavouring agent. It’s currently under testing as a skin penetration enhancer for the transdermal delivery of drugs. Studies focusing on nerolidol have shown antifungal, antibacterial, anxiolytic, and antioxidant properties.
NEROLIDOL AGAINST PARASITES AND INFECTIONS
Nerolidol has recently been studied for its antiparasitic, antifungal, and antibacterial properties. Research on both mice and cell cultures showed that nerolidol can reduce Leishmaniasis infection. This terpene inhibited the growth of parasites[1] in lab tests, and it may be further studied for the treatment of Leishmaniasis in humans, an infection afflicting 14 million people around the world, with a wide spectrum of diseases and resistance to therapeutic approaches.
Another study on animals found that nerolidol can reduce skin lesions[2] caused by the fungal infection from Microsporum gypseum, and it was also found that this terpene can work in synergy with antibiotics[3] to destroy bacterial pathogens more effectively than the single antibiotic drug. Other recent studies display the antiparasitic and insecticidal properties of this terpene, both on animals and plants.
One subject of interest to terpene researchers is malaria. The antimalarial activity of nerolidol was investigated[4] in a mouse model, where the terpene was delivered orally and via inhalation. In both cases, parasitaemia was inhibited by 80% up to almost 100% until 14 days after infection. After 30 days post-infection, the survival rate of orally treated mice was 90% compared to 16% of the non-treated mice. The toxicity of nerolidol was not significant.
NEROLIDOL AND THE BRAIN
Although it’s suspected that nerolidol does have mechanisms that affect the brain, it has not been explored like other essential oils for its anxiolytic effects on animal models of anxiety. However, a study investigated the effects on the central nervous system in mice, indicating that nerolidol exerts an anxiolytic effect[5] without altering motor coordination. The exact mechanisms of this action are still unknown, yet the lab results once again confirm the validity of the traditional use of certain fruits and herbs containing this terpene.
Another study[6] evaluated the neuroprotective effects of nerolidol against oxidative stress in neuronal cells compared to vitamin C. The research, carried out on mice, also evaluated the sedative effects of nerolidol compared to diazepam (first marketed as Valium). Nerolidol displayed sedative effects on animals, in addition to antioxidant effects on subjects’ hippocampus.
NEROLIDOL AND THE SKIN
One of most recognised effects of nerolidol is, as of today, the enhancement of dermal penetration by other active principles. Many terpenes are non-irritating transdermal penetration enhancers, and are therefore used in the pharmaceutical and nutraceutical industries. One study[7] showed a profound skin penetration enhancement effect made possible by terpenes. The rank order of the enhancement effect for diclofenac sodium with high doses of terpenes put nerolidol in first place, followed by farnesol, carvone, methone, and limonenoxide. The rank order with lower doses of terpenes was farnesol, carvone, nerolidol, menthone, and limonenoxide.
Users (and producers) of creams, lotions, and balms for the treatment of skin conditions, or simply for daily skincare, might consider this improved penetration of topicals made possible by nerolidol, as well as its non-toxicity, when choosing their preferred mix of ingredients.
NEROLIDOL IN THE CANNABIS PLANT
Terpene profiles can vary greatly in cannabis plants according to genetics and environmental factors. While it’s not so easy to pick a strain that contains a specific terpene, especially if it’s not one of the major ones, one hint is to consider that many cannabis strains that contain nerolidol might have a woody, fruity, and citrus aroma that persists in flower form and when combusted.
One example of a strain containing nerolidol is Royal Jack Automatic, which is an autoflowering version of the famous Jack Herer, with flavour and effects kept very close to the original. Another one is Royal Cookies, a variety that blends sweet and earthy tones, and measures up to 23% THC in its ripe buds. It’s good to remember that even the most persistent terpenes can degrade and burn out during combustion. Vaporising cannabis is always a better choice, both for your health and for maximising terpene expression. If you’re a grower, don’t forget to read our tips for maximising terpenes in your plants.
- Antileishmanial Activity of the Terpene Nerolidol | Antimicrobial Agents and Chemotherapy https://aac.asm.org
- Antifungal effect of eugenol and nerolidol against Microsporum gypseum in a guinea pig model. - PubMed - NCBI https://www.ncbi.nlm.nih.gov
- Sensitization of Staphylococcus aureus and Escherichia coli to Antibiotics by the Sesquiterpenoids Nerolidol, Farnesol, Bisabolol, and Apritone https://www.ncbi.nlm.nih.gov
- Antimalarial activity of the terpene nerolidol. - PubMed - NCBI https://www.ncbi.nlm.nih.gov
- Assessment of anxiolytic effect of nerolidol in mice https://www.ncbi.nlm.nih.gov
- Antioxidant effects of nerolidol in mice hippocampus after open field test. - PubMed - NCBI https://www.ncbi.nlm.nih.gov
- The effect of terpene concentrations on the skin penetration of diclofenac sodium. - PubMed - NCBI https://www.ncbi.nlm.nih.gov












































